Advancing medical training with augmented reality and haptic feedback simulator: outcomes of a randomized controlled trial on lumbar puncture | BMC Medical Education

Study design
This prospective, single center, parallel-group randomized clinical trial aimed at comparing the effectiveness and adding value of AR and haptic feedback simulation training with traditional bedside teaching for medical students learning to perform LPs.
Ethical approval for this study was granted by the Institutional Review Board (IRB) of the faculties of medicine, dentistry, nursing and midwifery on the 15th of October 2021 with the reference number CE-2021-128.
ClinicalTrials.gov ID NCT05269238, Registration Date 2022-01-14.
Participants
Medical students
Medical students in their clinical years, who had not previously performed an LP, were deemed eligible for inclusion. Students were recruited from the Rheumatology, Neurology and Neurosurgery departments at Strasbourg University Hospitals. Students who had previously performed an LP or an epidural infiltration were excluded from the study. All participating students provided written informed consent to participate in the study.
Patients
Adult patients requiring LPs as part of their routine clinical care were recruited from the same departments. Patients were excluded if they had a history of a previous LP, a Body Mass Index (BMI) greater than 35 kg/m², spinal ankylosis such as ankylosing spondylitis or Diffuse Idiopathic Skeletal Hyperostosis (DISH), inability to provide informed consent due to cognitive impairment or language barriers, were under legal protection, or had a contraindication to the use of EMLAPATCH 5%. Written informed consent was obtained from all participating patients.
Randomization
Patients were randomized in a 1:1 ratio to be managed by a student from either the simulation group or the control group, stratified by age, ensuring a balance between patients over and under 50 years of age. Medical students were also randomly assigned in a 1:1 ratio to either the simulation group or the control group using a block randomization method, with blocks of size four, stratified by department.
Interventions
Sim&Care simulator
The setup consists of a PC running the software, a Microsoft HoloLens 2® for the AR environment, a haptic interface (the Touch™ from 3D Systems) for force feedback, and a manikin serving as the physical support for AR (Fig. 1).
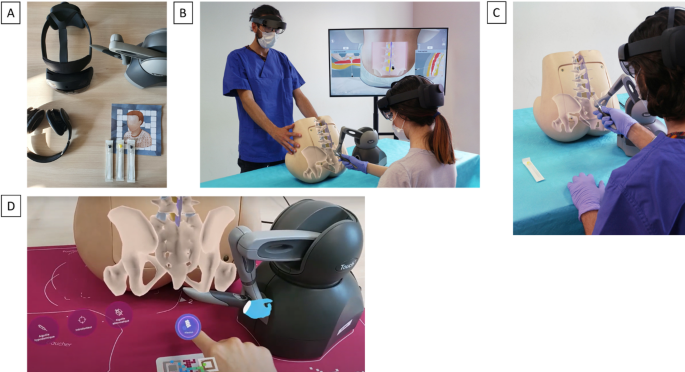
Simulator presentation. (A) Material provided for Sim&Care® simulation training. (B) Installation of the training session (C) Augmented reality seen through the Hololens 2® headset. (D) Haptic feedback material (Touch™ from 3D Systems)
Force feedback is generated through a real-time interactive biomechanical simulation, where each anatomical structure and medical instrument is modeled as a mechanical system, primarily using finite elements. When students manipulate virtual needles, the resulting interactions create contacts that are geometrically measured within the simulation. These contacts are integrated into the mechanical system as constraints, which account for the compliance of the simulated organs. The simulation then calculates the forces and friction associated with these interactions and transmits them at a high frequency to the haptic interface, generating realistic force feedback. This feedback is dynamically influenced by the mechanical properties of the contacting organs as well as the geometric and dynamic characteristics of the interaction.
Sim&Care® was developed by InSimo® in close collaboration with the medical team from the Rheumatology Department at Strasbourg University Hospitals. Each development step was carefully considered to ensure the tool met both medical and educational requirements. Repeated testing with medical students provided valuable feedback, refining the tool to better address user needs. AR was specifically chosen to replicate the LP environment and physician-patient interaction while emphasizing the educational goal of mastering the technical procedure. The development of the simulator, including its haptic and AR functionalities, occurred independently from this study and prior to its initiation. This study is the first to evaluate the educational impact of the simulator in a randomized controlled trial. None of the authors hold any financial interest in InSimo®, nor were the authors involved in the technical development or commercialization of the simulator. InSimo® provided technical support during the training sessions but was not involved in the study design, data collection, analysis, or interpretation. This delineation ensures the independence of the study and eliminates potential conflicts of interest.
Sim&Care® offers four different patient cases: standard, scoliosis, osteoarthritis, and a pediatric case (not used in this study). For each case, users can select one of two common LP positions: sitting or lateral decubitus. In this study, only the seated configuration was used during simulation training to maintain consistency across sessions. Although the lateral decubitus position is available within the simulator’s interface, it was not utilized during the training program. Various customization features enhance the simulation experience, including patient positioning, needle selection, and adjustable difficulty levels. Additionally, users can choose to display or hide internal anatomical structures in transparency and activate a visual aid that shows the needle’s trajectory, assisting in accurate needle placement within the spinal canal.
AR simulation group
Students in the simulation group received a combination of standard theoretical training on LP procedures; a classical bedside training for at least one LP and a 2-hour practical training session using the Sim&Care® simulator within the first 5 days of their hospital rotation. The session followed a structured curriculum provided by Sim&Care®, incorporating guided practice with an expert instructor (RF) and a standardized set of LP tasks. The simulator integrates interactive educational content—such as patient positioning, needle selection, and difficulty adjustment—alongside guidance from both technical and medical instructors. However, it does not replace learning with an experienced physician. A doctor is always present to explain the procedure beforehand and provide medical instructions before students use the simulator. During the session, an InSimo® technical trainer assisted students, ensuring they could focus on mastering the technique rather than managing the simulator. Each student followed the same structured learning sequence: beginning with an LP on a standard patient with all available aids, then progressing to more complex cases (scoliosis or osteoarthritis), and finally tackling the most challenging scenario, where augmented reality anatomical visualization was disabled.
Control group
Students in the control group received the same standard theoretical training on LP procedures followed by bedside training. This training included observation and hands-on practice under the supervision of experienced practitioners for at least one LP before performing it alone on a patient.
At the end of their clinical placements in the corresponding hospital departments, the students in the control group also benefited, if they so wished, from a training session with the simulator. This ended their participation in the study.
Procedure
Students were included and randomized at the start of their rotation. Once a patient had been identified, included and randomized, a student from the corresponding department and from the corresponding randomization group (simulator or control) was assigned to perform the LP. If no student from the corresponding department was available, then a student from another department but from the corresponding randomization group was assigned. If no student from the corresponding randomization group was available, the LP was perfomed out-of-protocol.
In both groups, the supervising physician determined the choice of needle, sanitization protocol, and patient positioning, which could be either seated or lying down. The LP was performed under the supervision of a senior physician who could intervene if necessary. If the student was unsuccessful in obtaining cerebrospinal fluid (CSF) on the first attempt, they were allowed to reposition the needle or use a new needle, provided the patient consented. The procedure was terminated if deemed too lengthy or unsuccessful by the supervising clinician.
Outcome measures
Primary outcome
The primary outcome was the success rate of LPs, defined as the successful collection of CSF without assistance or interruption by the patient or the investigator.
Secondary outcomes
Secondary outcomes included patient pain and patient anxiety, assessed using self-defined Likert scales from 0 to 10, immediately after the procedure (Supplementary file); the duration of the procedure measured in seconds from the start of patient setup to the collection of the first drop of CSF; the incidence of traumatic LPs, defined as CSF with more than 10 red blood cells per cubic millimeter, and the occurrence of post-LP syndromes. We also assessed student comfort and anxiety, self-assessed using Likert scales from 0 to 10 for comfort during the procedure, material preparation, patient positioning, sanitization, and communication with the patient (Supplementary file). The anchor descriptions for these scales were as follows: 0 = not at all / none, 5 = moderate / neutral, and 10 = extremely / completely. For pain-related questions, 0 represented no pain at all, while 10 indicated the worst pain imaginable. Similarly, for confidence and ease-of-use measures, 0 denoted no confidence or extreme difficulty, and 10 represented complete confidence or ease.
Exploratory outcomes
We evaluated the pedagogic and material aspects of the training from the students’ perspectives. This included a detailed assessment of their experiences with Sim&Care® simulator. Upon completion of their simulator training program, students were asked to complete a comprehensive questionnaire designed to measure various aspects of their training experience (Supplementary file). This questionnaire included items on pedagogic evaluation, material evaluation, organization of training, the added value of the training compared to theoretical instruction, the realism of the experience, the perceived added value of haptic feedback and the AR headset, emotional difficulty, and their comfort level for performing future LPs. Each item was rated on a 0–10 scale(0 = not at all / none, 5 = moderate / neutral, and 10 = extremely / completely). For the simulation group, this evaluation took place immediately after the simulator training session, at the beginning of their participation in the study. For the control group, the simulator training and the evaluation occurred at the end of their participation in the study.
Statistical analysis
The sample size calculation was based on the primary endpoint, which assesses the proportion of successful LPs in each arm (as defined previously). It was impossible to establish a success rate for LP after simulator training, as the simulator had never been evaluated before. The available data literature about first LP success rate is scarce and this parameter can be highly variable between countries and health systems. We used the success rates from a unpublished French medical thesis comparing theoretical training with training using a procedural simulator [10]. This study reported a success rate of 30% when performing a first LP in the absence of specific training other than theoretical training, and 68% after training with a back procedural simulator. Given that, a minimum number of 26 patients per group allowed to achieve a power of 80% (alpha = 0.05). This number was increased to 30 to maintain power close to 80%, even if the success rate turned out to be slightly lower than reported and to compensate for possible withdrawals from the study.
Data were analyzed using univariate methods. Continuous variables were presented as mean ± standard deviation (SD) or median and interquartile range (IQR) depending on the distribution. Normality was assessed using the Shapiro-Wilk test. Categorical variables were summarized as frequencies and percentages. The primary outcome was analyzed using Fisher exact test. Significance of the differences between groups for the secondary outcome analyses were evaluated using the Student t-test, Chi-square test or Fisher’s exact test, as appropriate. Data were analyzed on an intention-to-treat basis, including all randomized participants. Statistical analyses were conducted using R [11] version 4.4.1. R analysis scripts are available online at All tests were two-sided, and a p-value of < 0.05 was considered statistically significant.
As specified in the study protocol, an interim analysis was planned and performed after enrolling at least 40 patients of the target sample size. A predictive probability calculation was implemented to determine the likelihood of achieving the primary endpoint at study completion, conditional on the data observed at the interim timepoint. This approach allows for estimating the probability of ultimate study success based on the accumulated data from the first 40 participants, while accounting for the uncertainty in the outcomes of the remaining participants yet to be enrolled.
link





![Locum tenens offers physicians a path to freedom [PODCAST] Locum tenens offers physicians a path to freedom [PODCAST]](https://kevinmd.com/wp-content/uploads/Design-4-scaled.jpg)
